Segment SwitzerlandMARKET ENVIRONMENTBUSINESS PERFORMANCE
2020 was marked by the global pandemic caused by the spread of COVID-19. As part of the federal scheme for continuity of supply of therapeutic products, Zur Rose worked closely with the authorities to ensure the best possible security of supply of medicines to the population. The coronavirus crisis clearly revealed that the digitalisation of Swiss healthcare is lagging and there is an urgent need for modernisation.
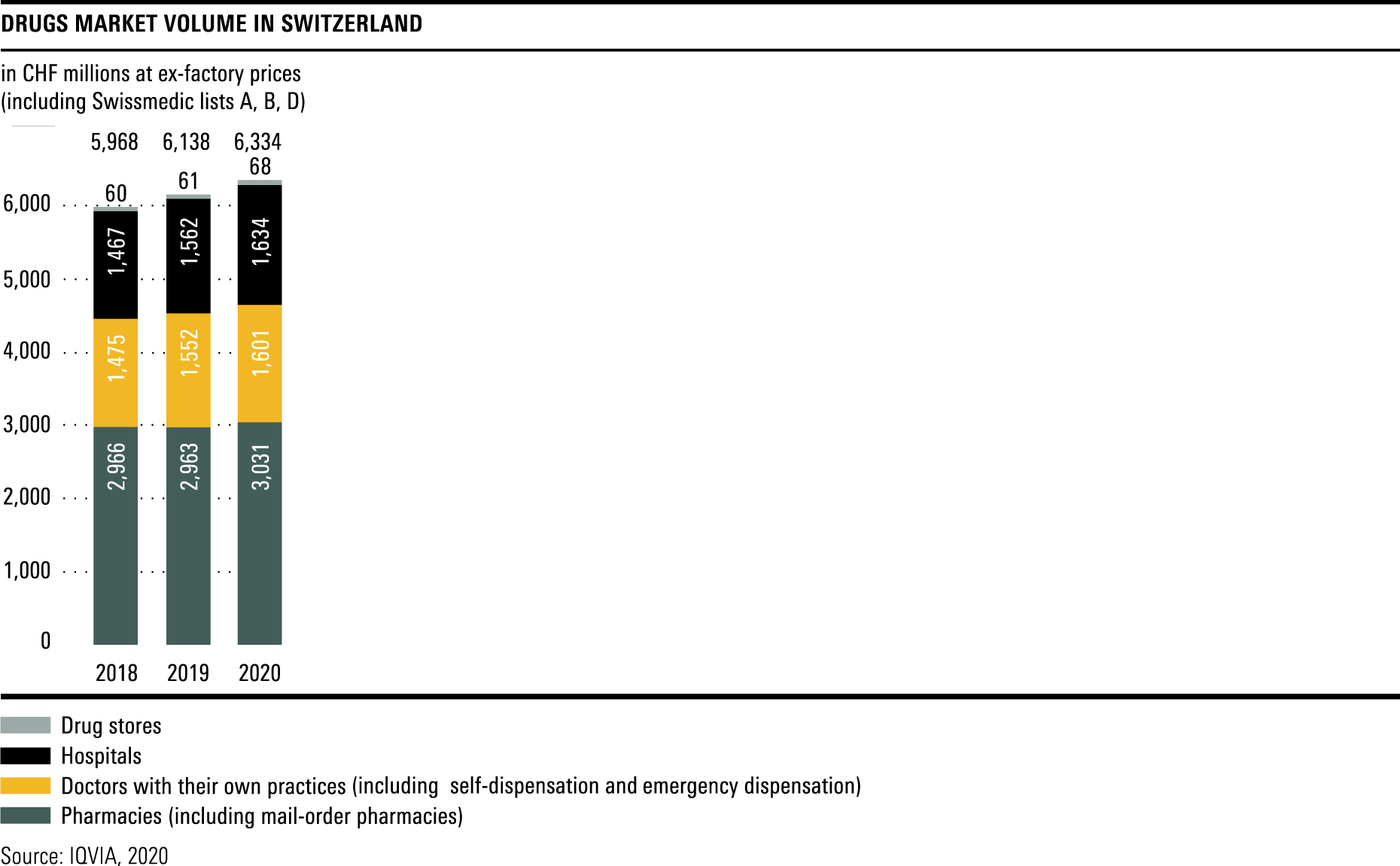
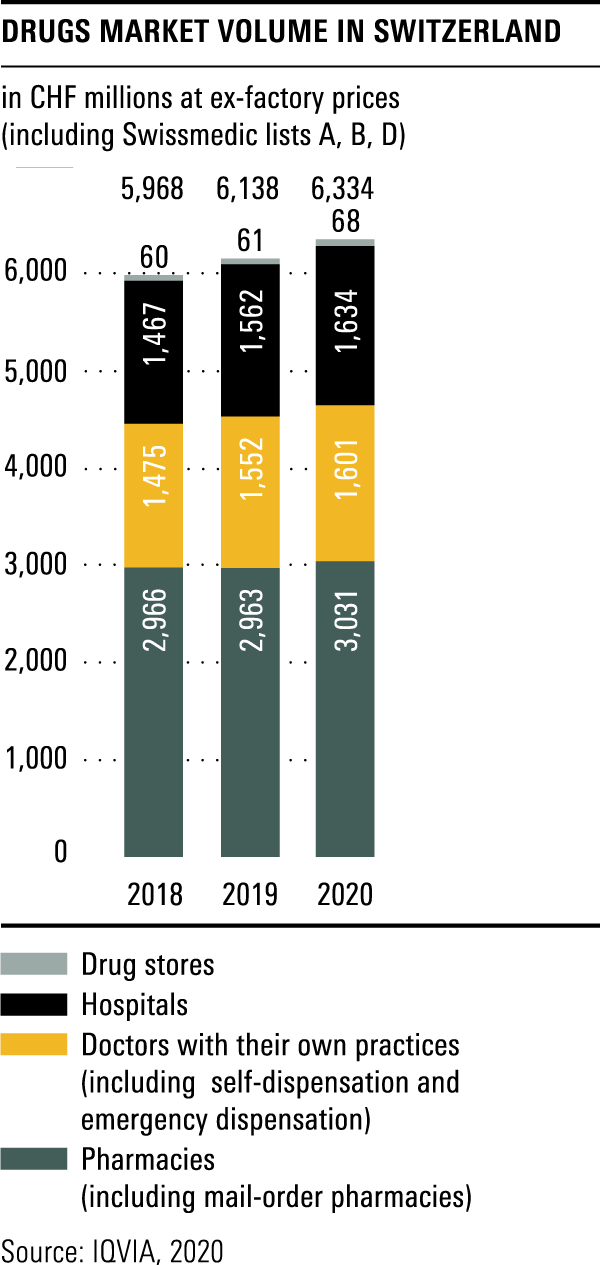
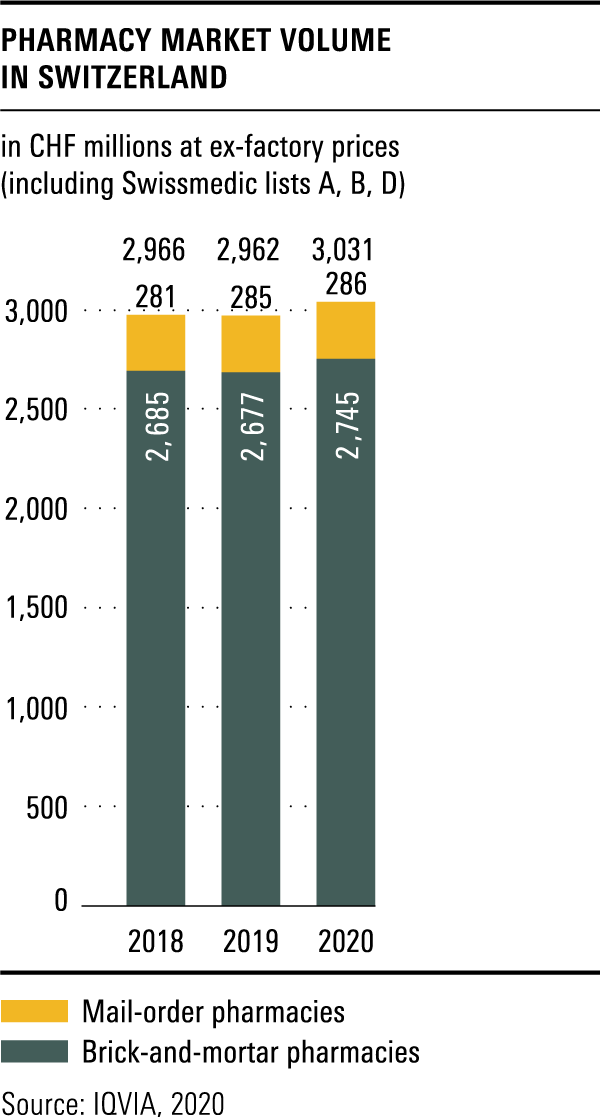
Trends in the market for medicines
The total volume in the drugs market in 2020 amounted to CHF 6.3 billion, a year-on-year rise of over 3 per cent.
Application for exemption permit to operate a mail-order service for non-prescription emergency, cold and flu medicines during the pandemic
As part of the federal scheme for continuity of supply of therapeutic products, Zur Rose has a responsibility to ensure the best possible security of delivery and supply of medicines to the population. Since the beginning of the pandemic customers have been increasingly asking for the ability to also order non-prescription drugs online; this is still not permitted under Swiss law at present. At the same time, the population has been encouraged to stay at home owing to the risk of infection.
On 23 March 2020, Zur Rose therefore submitted an application to the Federal Council and the Federal Office of Public Health (FOPH) for a temporary exemption permit to sell non-prescription drugs for self-medication of the symptoms of flu and colds by mail-order. For reasons we find hard to understand, in a letter dated 16 April Health Minister Alain Berset and the FOPH declined this request, arguing that the authorities saw no reason for emergency provisions to suspend the ban on selling non-prescription medicines by mail-order. They claimed arrangements for supplying the population with medicines were functioning well, not least because bricks-and-mortar pharmacies and drugstores were carrying out home deliveries.
In June National Councillor Marcel Dobler raised the issue in an interpellation, asking how the government explained the fact that mail- order pharmacies were unable to deliver OTC medications to customers without a prescription, whereas bricks-and-mortar pharmacies were allowed to provide the same services in the form of home delivery. The response from the Federal Council on 19 August was as follows: “The Federal Council acknowledges that in practice the current regulations governing home delivery of medicines results in disparities compared to the rules governing the mail-order business.” It continued: “The Federal Council is willing to examine better ways of distinguishing between mail-order and home delivery at the federal level with a view to avoiding unwanted distortions in competition.”
Accelerating the digitalisation of healthcare
The coronavirus pandemic clearly revealed how digitalisation in healthcare is lagging: media breaks and the absence of digitalised processes made crisis management more difficult and prevented data from being transmitted efficiently. The call for more digitalisation in healthcare grew louder. Electronic prescriptions are a key part of this. Two motions in the federal parliament during the year under review requested that these also be brought in across Switzerland. The benefits for patients are obvious: electronic prescriptions guarantee legibility, thus increasing patient safety and cutting the follow-up costs due to incorrect medication. Zur Rose is committed to the mandatory introduction of electronic prescriptions in Switzerland.
Fair system for drug prices
In its debate on cost-cutting measures for the healthcare system at a special session early in the year, the National Council rejected the introduction of a reference price system for patent-expired drugs by a clear majority of over two-thirds. Although regulated prices for prescription medicines and generics have already been regularly reviewed and reduced in the past, the package of cost-cutting measures also provides for new arrangements for regulating prices and hence margins on medicines. Zur Rose welcomes the rejection of the reference price system and emphasises that any restructuring of pricing cannot regulate individual elements in isolation; factors such as ex-factory and generics prices, service-based remuneration and distribution margins have to be seen as an inter-related whole. Redefinition must involve all relevant players, especially also to ensure provision of medicines to tpeople living with a chronic condition who often depend on drugs in the medium to high-price segment.